Pfizer officially asks the FDA to authorize its COVID vaccine for kids aged 5-11

Enlarge this image
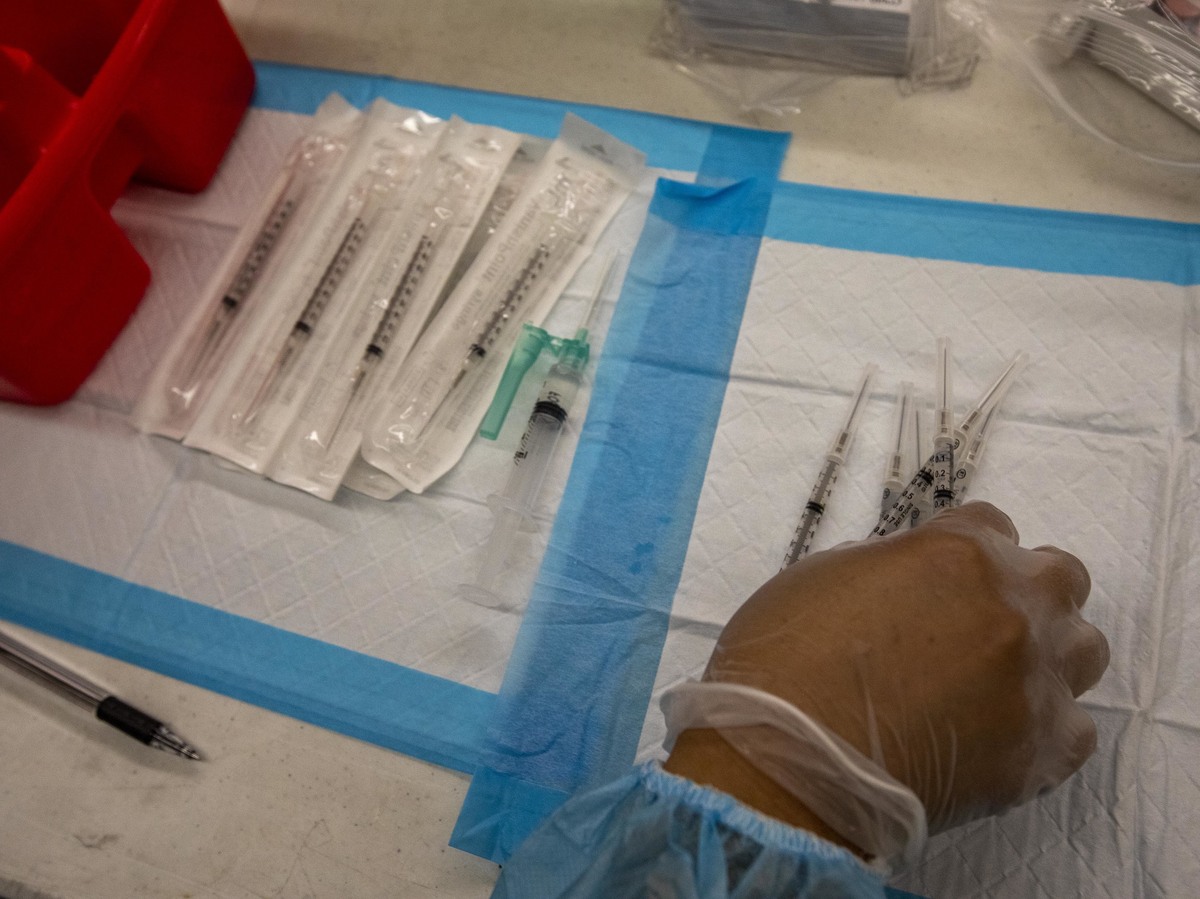
A healthcare worker prepares doses of the Pfizer-BioNTech Covid-19 vaccine at a mobile vaccination clinic at a high school in Los Angeles in August.
Jill Connelly/Bloomberg via Getty Images
hide caption
toggle caption
Jill Connelly/Bloomberg via Getty Images
A healthcare worker prepares doses of the Pfizer-BioNTech Covid-19 vaccine at a mobile vaccination clinic at a high school in Los Angeles in August.
Jill Connelly/Bloomberg via Getty Images
Pfizer and BioNTech are officially asking the Biden administration to authorize the use of their COVID-19 vaccine for children ages 5 to 11.
Pfizer tweeted on Thursday that the companies had submitted their formal request for Emergency Use Authorization of the vaccine to the Food and Drug Administration.
«With new cases in children in the U.S. continuing to be at a high level, this submission is an important step in our ongoing effort against #COVID19,» the pharmaceutical giant said.
Currently, the vaccine is only in use in the U.S. for people ages 12 and older.
- pfizer
- vaccine













Комментарии 0